- Research
Published:
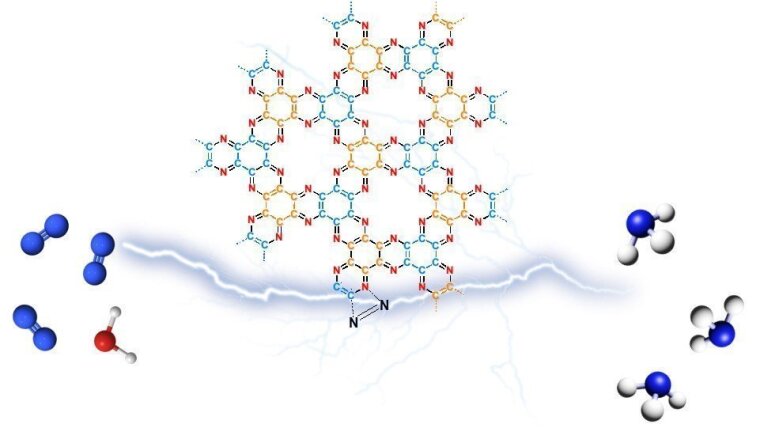
The electrochemical nitrogen reduction reaction (NRR) to ammonia (NH3) is a potentially carbon-neutral and decentralized supplement to the established Haber–Bosch process. Catalytic activation of the highly stable dinitrogen molecules remains a great challenge. Especially metal-free nitrogen-doped carbon catalysts do not often reach the desired selectivity and ammonia production rates due to their low concentration of NRR active sites and possible instability of heteroatoms under electrochemical potential, which can even contribute to false positive results. In this context, the electrochemical activation of nitrogen-doped carbon electrocatalysts is an attractive, but not yet established method to create NRR catalytic sites. Herein, a metal-free C2N material (HAT-700) is electrochemically etched prior to application in NRR to form active edge-sites originating from the removal of terminal nitrile groups. Resulting activated metal-free HAT-700-A shows remarkable catalytic activity in electrochemical nitrogen fixation with a maximum Faradaic efficiency of 11.4% and NH3 yield of 5.86 µg mg−1cat h−1. Experimental results and theoretical calculations are combined, and it is proposed that carbon radicals formed during activation together with adjacent pyridinic nitrogen atoms play a crucial role in nitrogen adsorption and activation. The results demonstrate the possibility to create catalytically active sites on purpose by etching labile functional groups prior to NRR.